Water
![]()
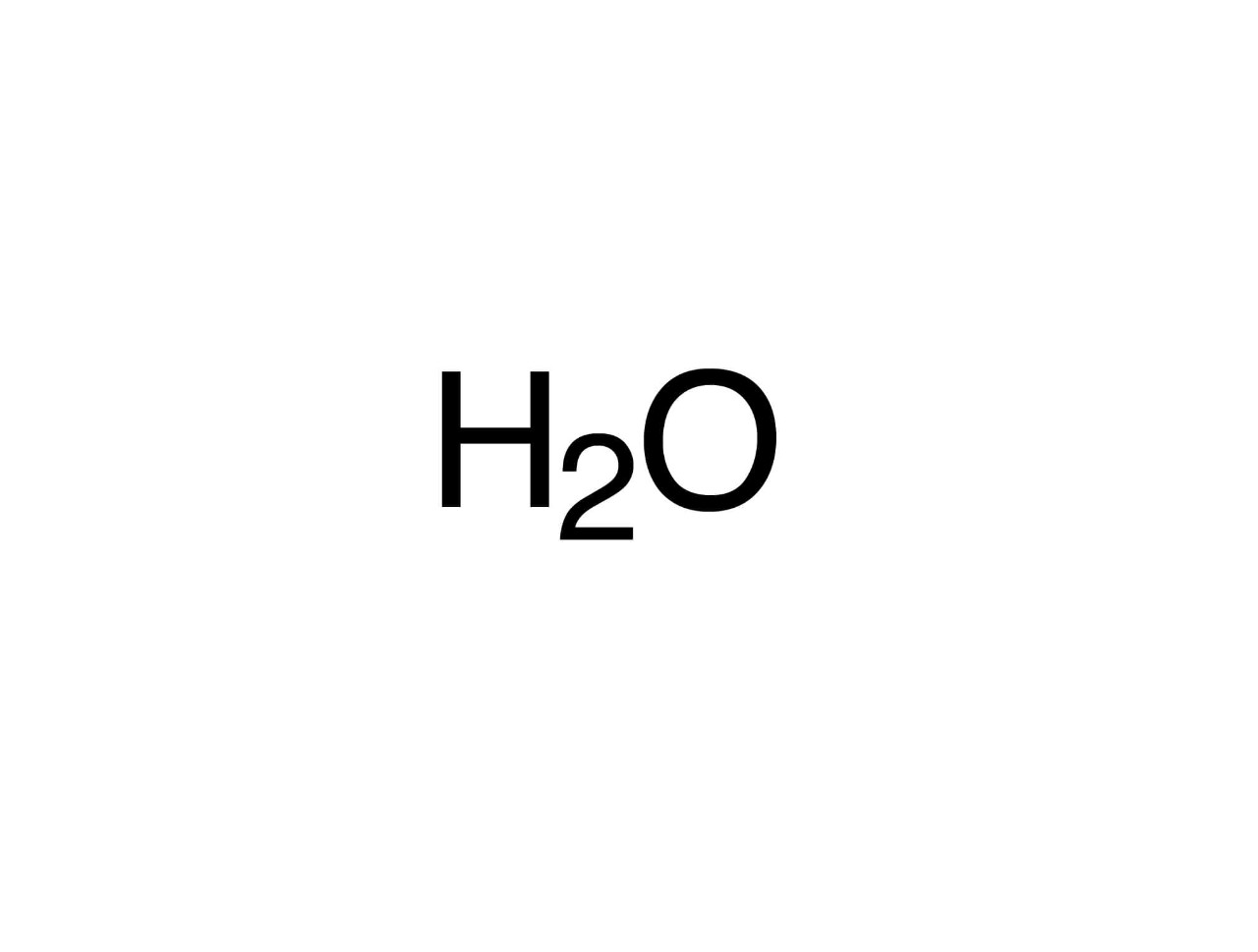
Known as the “universal solvent,” water (H2O), CAS 7732-18-5, covers 71 percent of the Earth and is essential to human life. It is a clear, odorless, tasteless liquid at ambient temperature; it freezes to ice below 32°F (0°C) and boils above 212°F (100°C). Ice is also colorless in appearance, while water vapor is essentially invisible.
Water for laboratory use is available in specific grades based on purity:
- Ultrapure — for use in HPLC, AAS, and other applications
- Purified — for use in the preparation of samples and buffers
- Primary grade — for use in water baths, autoclaves, and cleaning
Application-specific water includes analytical water, chromatography water, and life sciences water (for use in cell culture, molecular biology, and biotechnology). Water has myriad other applications in science and industry as a solvent or reactant and, less commonly, as a catalyst.
While water does not pose any hazards to the human body, water for laboratory use should never be ingested.


